Dive Brief:
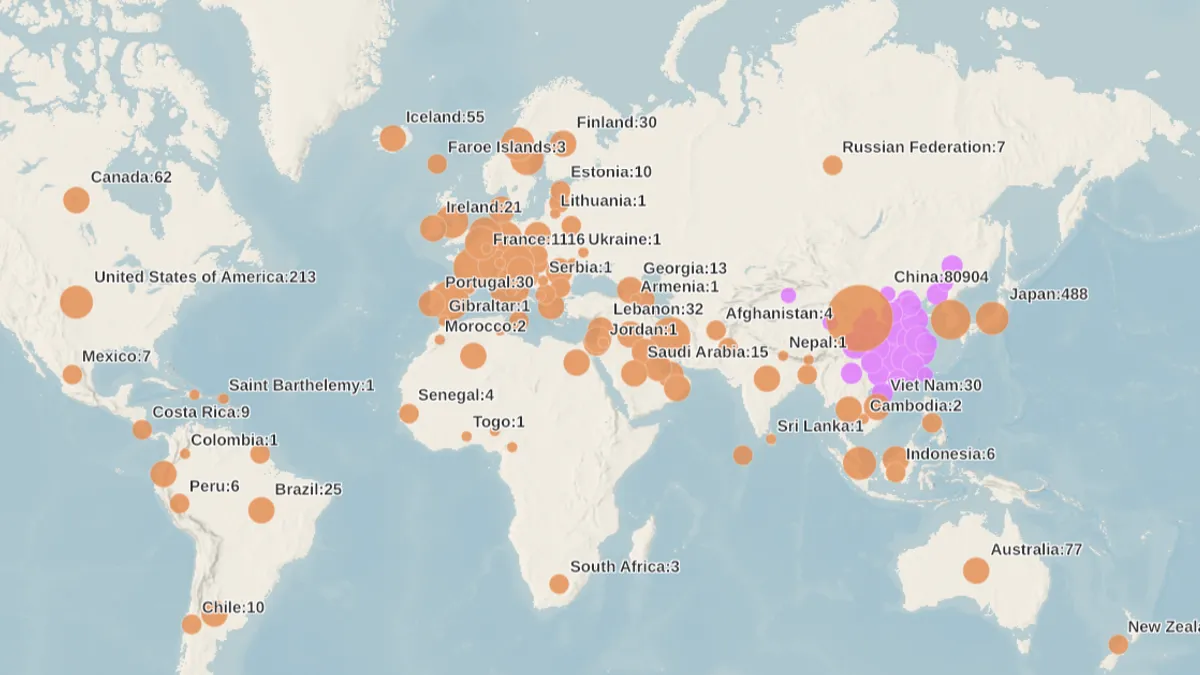
- Hospitals in some of the areas hardest hit by COVID-19 have started to dip into emergency supplies for personal protective equipment. The World Health Organization has said the situation is "very close" to a pandemic as more than 113,600 cases were reported worldwide as of late Tuesday morning.
- The Joint Commission on Monday said it had canceled surveys in the Seattle area indefinitely "because of the evidence of sustained community spread" of the virus. It clarified that surveys elsewhere will have an intensified focus on infection control processes and emergency management planning. The accrediting organization has also suspended travel to several countries.
- Vice President Mike Pence, who is leading the administration's response to the outbreak, said Monday evening White House officials would be meeting with hospital and health insurance leaders, although he gave no further details. Healthcare Dive confirmed an executive from Aetna would be attending a meeting at the White House on Tuesday. Other payers did not respond to requests for comment.
Dive Insight:
The U.S. response to COVID-19 continues to accelerate. Across the country, schools are closing and large gatherings are being called off. Several federal lawmakers are in self-quarantine after contact with people who have shown symptoms of the disease and stock markets tanked drastically as the work week kicked off.
Payers are ramping up attempts to improve access to testing and treatment for coronavirus. Humana said Monday it would not charge for urgent care telemedicine visits for the next 90 days. Aetna has waived cost-sharing for virtual visits for the same time period and UnitedHealth Group is encouraging telehealth use.
The moves came days after an $8.3 billion coronavirus funding bill loosened Medicare regulations so that more people could access telehealth treatment.
On Monday, CVS Health said it would waive charges for home delivery of prescription medications and early refill limits on 30-day prescription maintenance medicines for all CVS Caremark members. That comes as public health officials suggest people at high risk for being affected by coronavirus keep extra supplies of needed medication on hand.
CMS late Monday urged hospital emergency departments to redouble their infection control and prevention policies and reminded facilties they should immediately contact local public health officials if a patient who comes to the ER is suspected of having COVID-19. "Sending and receiving hospitals have an obligation to follow appropriate procedures for the safety of all involved," the agency said.
Hospitals throughout the country are bracing for a wave of patients.
Jan Emerson-Shea, vice president of external affairs for the California Hospital Association, told Healthcare Dive the group had heard some hospitals were getting low on PPE and masks in particular.
Hospitals in that situation can contact their local county health department or EMS agency for help acquiring additional supplies. The California Department of Public Health has made available some of the 21 million N95 masks it has on reserve to help ease shortages.
A spokesperson for the Greater New York Hospital Association said none of its members had reported a shortage. but said some "have obtained PPE from New York City's ample emergency stockpile."
Earlier federal actions have allowed for masks not officially approved for healthcare settings to be used by health professionals and HHS is buying 500 million N95 masks over the next 18 months for the Strategic National Stockpile.
HHS Secretary Alex Azar told the American Association of Nurse Practitioners on Tuesday the department is "keenly focused on making sure that frontline healthcare responders like you have the protective equipment you need for treating patients," according to prepared remarks.
Hospitals have adapted in other ways as well. The University of Washington Medical Center in Seattle created a drive-thru testing clinic for its employees by repurposing an on-site garage, while Intermountain Healthcare restricted hospital visitations. Multiple organizations have limited employee travel.
The industry has been affected in other ways as well. Numerous conferences and medical meetings have been canceled or postponed. Outbreaks at some pharma companies have prompted them to mandate work-from-home policies.
Also, the FDA has cracked down on fraudulent products purporting to cure COVID-19, which has no vaccine yet.